News Archive
RESEARCH HIGHLIGHT - Distinct alpha-Synuclein species induced by seeding are selectively cleared by the Lysosome or the Proteasome in neuronally differentiated SH-SY5Y cells
IMPRiND partners BRFAA and CNRS established a cell model to assess aggregation and turnover of α-Synuclein (α-Syn) assemblies. Reporting in Journal of Neurochemistry, Pantazopoulou et al. used neuronally differentiated SH-SY5Y neuroblastoma cells with inducible expression of α-Syn and induced seeding of endogenous Proteinase K-resistant α-Syn species with the addition of α-Syn pre-formed fibrils (PFFs). Subsequently, the shutting down of the expression of α-Syn in this inducible cell model resulted in the clearance of α-Syn aggregates. Using this model, it was demonstrated that macroautophagy seems to serve as the major pathway for clearance of highly aggregated α-Syn assemblies whereas the Proteasome System is implicated in the degradation of phosphorylated at S129 α-Syn oligomers. These findings that different degradation pathways induce the clearance of distinct α-Syn aggregated species represent new and important insights into the biology of α-Syn aggregation and turnover.
This well established cell model can prove an essential tool to assess aggregation and turnover of α-Syn assemblies as well as the role of different post-translational modifications (i.e. phosphorylation, ubiquitylation, truncation, sumoylation) and their effect on oligomerization, and to further screen for modifiers affecting α-Syn aggregation, clearance, secretion and cell-to-cell transmission. A deeper understanding of the mechanisms underlying aggregation propensity and clearance may help design novel strategies for regulating the levels of toxic α-Syn conformers and eventually develop a treatment for PD and related synucleinopathies.
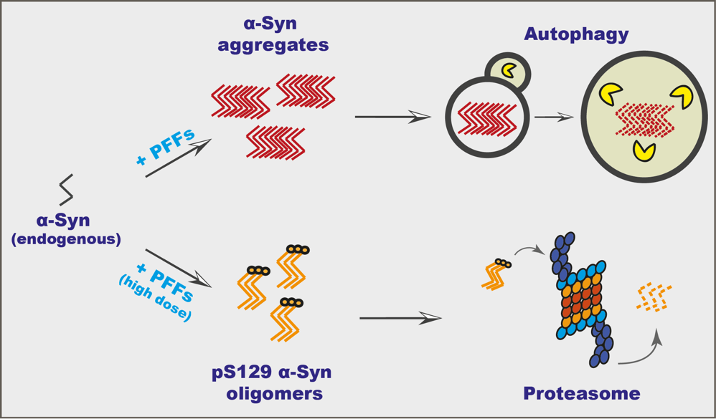
Different degradation pathways induce the clearance of distinct α-Syn aggregated species. Upon incubation with recombinant fibrils (PFFs), endogenous α-Syn aggregates are formed and Autophagy serves as the major pathway for clearance. pS129 α-Syn oligomers are apparent only upon incubation with high-dose PFFs, and the Proteasome system is implicated in the degradation process.
This project receives funding from the Innovative Medicines Initiative 2 Joint Undertaking (www.imi.europa.eu) under grant agreement No 116060. This Joint Undertaking receives support from the European Union’s Horizon 2020 research and innovation programme and EFPIA.
This work is supported by the Swiss State Secretariat for Education‚ Research and Innovation (SERI) under contract number 17.00038.
The opinions expressed and arguments employed herein do not necessarily reflect the official views of these funding bodies.



